Key Takeaways:
In today’s beauty landscape, aspiration alone no longer sells products. Consumers want evidence, retailers want substantiation, and increasingly, founders are discovering that glossy claims without defensible data carry more risk than reward.
This shift was the focus of BeautyMatter’s recent webinar, Raising the Bar on the Skincare Game, which examined the evolving role of consumer and clinical testing in beauty. Moderated by BeautyMatter founder and CEO Kelly Kovack, the conversation brought together Elizabeth Corrigan, founder and CEO of Prestige Testing Agency; Yovanella Lemaitre, Vice President, International, at Circadia; and Neelam Muizzuddin, PhD, President of Skin Clinical Research Consultants.
“The modern consumer’s basket is not defined by impulse, but by validation,” Kovack said in opening the discussion. “Functional benefits are expected, and proof elevates the brand into the realm of trust.”
Proof Is Imperitive
Panelists agreed that the expectation for clinical evidence has intensified across every level of the industry—from investors and retailers to consumers themselves.
“Consumers are demanding proof-of-concept efficacy for them to make their decision-making process,” said Corrigan. She pointed to the data underscoring the shift: “92% of consumers prioritize clinical testing as part of their decision-making process.”
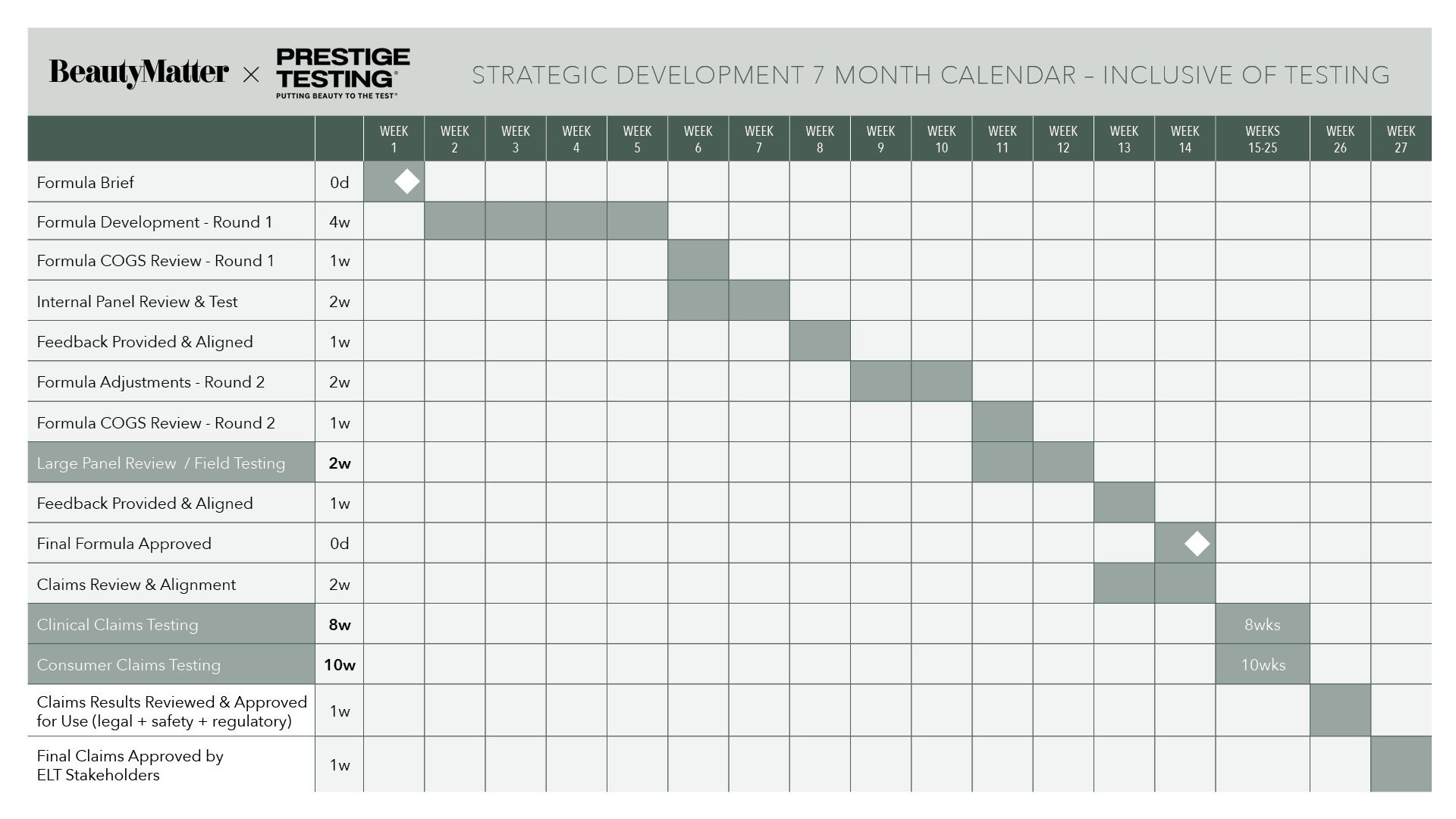
That demand is also pushing brands to rethink when and how testing happens. Historically, testing often occurred late in product development in order to generate marketing claims. Increasingly, brands are integrating testing earlier in the process.
Corrigan described the growing use of field testing during development to identify both adoption drivers and potential barriers before a product launches. “You want to understand early what is driving either adoption of the product or rejection of something about this product,” she said. “Fail fast, pivot, and tweak during the development process.”
The industry, she noted, is also becoming more sophisticated about understanding its audience. “I’m seeing customer insight divisions being built up within internal organizations,” Corrigan said. “The days of being in an echo chamber and speaking to yourself about who you think your customer is and what they want is finally starting to leave our industry.”
The Science Behind the Claims
As claims grow more complex, the science required to support them is becoming more demanding as well.
Muizzuddin explained that designing credible clinical study begins with defining the precise biological outcome a brand hopes to demonstrate. “To design a study, first of course we need to know what the objective is,” she said.
Some endpoints are relatively straightforward to measure. Cell turnover, for example, can be assessed through staining techniques that track how quickly skin sheds. “The epidermis constantly differentiates,” Muizzuddin said, describing how viable skin cells eventually become corneocytes on the surface of the skin. “We can stain the skin and observe how the color disappears as the skin sloughs off.”
Claims around collagen, however, are significantly more complex. Because collagen resides in the dermis—below the epidermis—measuring its changes requires more advanced technologies.
“Collagen is a slow process,” Muizzuddin explained. “We have to make sure the study runs long enough to detect meaningful changes.”
Researchers may use imaging technologies such as ultrasound or confocal microscopy to evaluate dermal density and structure. Other approaches measure indirect indications of collagen activity, such as improvements in elasticity and firmness.
“Instruments can displace the skin and observe how quickly it returns to its original position,” Muizzuddin said. Devices like the Cutometer, Ballistometer, and Twistometer help researchers quantify elasticity and firmness—two key proxies of dermal health.
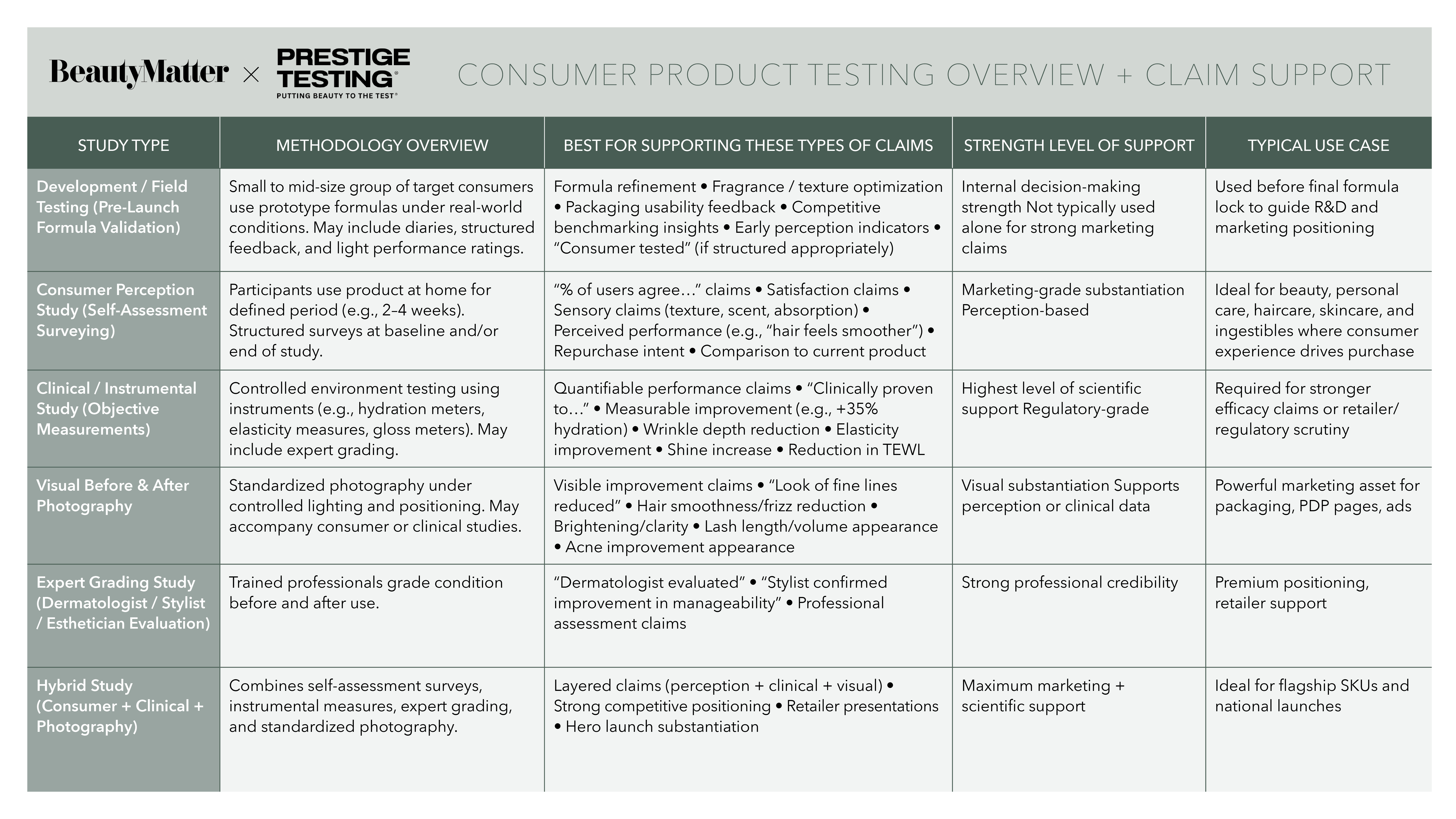
To fully understand product performance, Muizzuddin emphasized that multiple data sources are necessary. “Photographs show visible changes. Instruments detect changes you may not see visually. The subject's own experience is something that instruments cannot capture,” she added. “We need all three to get a full picture of what the product is doing.”
Clinical Evidence Meets Consumer Experience
For brands operating in professional channels, the need for substantiated results is particularly pronounced. Circadia, which sells exclusively through estheticians and skin professionals, has seen growing demand for clinical validation from its practitioner network.
“Estheticians are demanding more clinical studies,” said Lemaitre. “It gives our professionals measurable proof that they can share with confidence with their clients.”
Rather than focusing solely on individual product claims, Circadia chose to test entire treatment protocols. “We focused on testing a complete routine of products rather than an individual product,” she said. That approach supports both retail recommendations and in-cabin treatments.
At the same time, Circadia paired instrumental testing with consumer perception studies. The goal was to capture not only measurable results but also the emotional and sensory aspects of the treatment experience.
“Results are essential,” Lemaitre said. “But the experience of both the consumer and the esthetician is really important.”
Consumer perception studies help measure factors like overall satisfaction, confidence in results, and sensory experience. “It helps us validate how the consumer felt when receiving the treatment protocol,” said Lemaitre.
That balance between efficacy and experience is increasingly critical in a market where performance alone may not guarantee repeat purchase.
The Challenge of Authentic Visual Proof
Visual documentation remains one of the most powerful—and most misunderstood—forms of proof. Corrigan noted that many brands struggle to distinguish between marketing imagery and clinically valid before-and-after photography. “Marketing-facing photography is very different from true before-and-after photography,” she said.
Clinical photography requires strict standardization in lighting, positioning, and imaging conditions. While the results may not always be visually flattering, authenticity is precisely what builds credibility. “It is not the prettiest,” Corrigan acknowledged. “But it’s authentic.”
Muizzuddin echoed the importance of rigor. Poorly executed imagery can undermine otherwise valid results. “The photographs have to be very precise,” she said. “You should be able to see all the pores and match the exact area.”
At the same time, the rise of AI is adding another layer of complexity to visual proof. As generative tools make image manipulation easier and more convincing, brands will likely face increasing pressure to demonstrate that clinical imagery has not been digitally altered.
Corrigan noted that authentic, clinically captured photography will become even more important as trust becomes harder to establish in an AI-saturated environment. The industry may ultimately need clearer standards around what constitutes verifiable visual evidence, particularly as consumers grow more skeptical of images that appear overly polished or manipulated.
For brands, the message is clear: credible imagery, supported by transparent testing protocols, is becoming a critical component of building consumer trust.
Navigating the Longevity Conversation
As the industry increasingly embraces the language of longevity, panelists cautioned that marketing enthusiasm must be matched with scientific discipline. The new terminology may reflect a broader wellness narrative, but the biological processes involved remain complex.
“In terms of scientific validation, we may need to move toward slightly invasive testing,” Muizzuddin said. Biopsies and molecular analysis can provide deeper insight into processes such as cellular senescence or collagen remodelling.
Researchers may examine biomarkers such as telomere length—DNA sequences that shorten with age—or gene expression patterns to evaluate biological changes.
The most widely used approach today is gene profiling, Muizzuddin explained. By analyzing which genes are unregulated or downregulated after treatment, researchers can build a molecular profile of how a product interacts with the skin.
Even so, Corrigan cautioned brands against rushing into aggressive claims. “Collagen is one area where everybody needs to take a beat,” she said. “The last thing you want to do is overpromise and underdeliver.”
The GLP-1 Effect
Another emerging factor reshaping testing protocols is the rise of GLP-1 medications for weight loss.
Rapid weight loss associated with these drugs can produce visible changes in skin volume, elasticity, and hair density, creating new opportunities and challenges for skincare brands.
Corrigan said testing panels are now beginning to segment GLP-1 users from non-users to better understand how products perform across different physiological conditions. “You can’t assume a product will behave the same way for someone experiencing significant volume loss.”
Circadia has responded by developing treatment protocols that complement medical weight-loss journeys. “We suggest pairing longevity treatment protocols with GLP-1 treatments to help address firmness and hydration,” Lemaitre said.
Testing for Every Stage of Brand Growth
While clinical testing can sound expensive, with full clinical studies reaching $40,000 or more, panelists emphasized that brands can begin with smaller, more targeted studies.
Hydration measurements paired with consumer perception studies, for example, can offer meaningful validation at lower cost.
“Start simple,” Corrigan advised. “Do a hydration reading and a consumer perception study. Then build from there.”
Ultimately, testing is becoming less about compliance and more about competitive advantage.
As Kovack noted during the conversation, “Proof is the most defensible advantage that a brand can have.”
In a category increasingly shaped by data, transparency, and increasingly informed consumers, that proof may soon be the difference between a compelling claim and a credible one.